Design personalized CRFs effortlessly, without any technical background
Adapt your Electronic Case Report Forms to meet your needs without depending on IT experts.
Improving the capture, sharing and reuse of clinical research data within Biomedical Research Institutes through the use of Semantic Case Report Forms (CRF).
Adapt your Electronic Case Report Forms to meet your needs without depending on IT experts.
Advanced management and analysis of patient recruitment results using semantic technologies.
An RDF- and OWL-based platform designed to handle large volumes of clinical data and evolving research environments.
Leverage fields and concepts defined in other projects to promote efficiency and consistency in your studies.
View and analyze clinical data using semantic tools without needing to be proficient in RDF or OWL.
PATIENTS RECRUITED
RESEARCH PROJECTS
REPORTS REGISTERED
DATA FIELDS DEFINED
DEFINED REPORT TYPES
WORKFLOW STAGES
CURRENT USERS
Collect patient data effortlessly — designed for researchers, simple for participants.
Perfect for clinical and academic studies of any size.
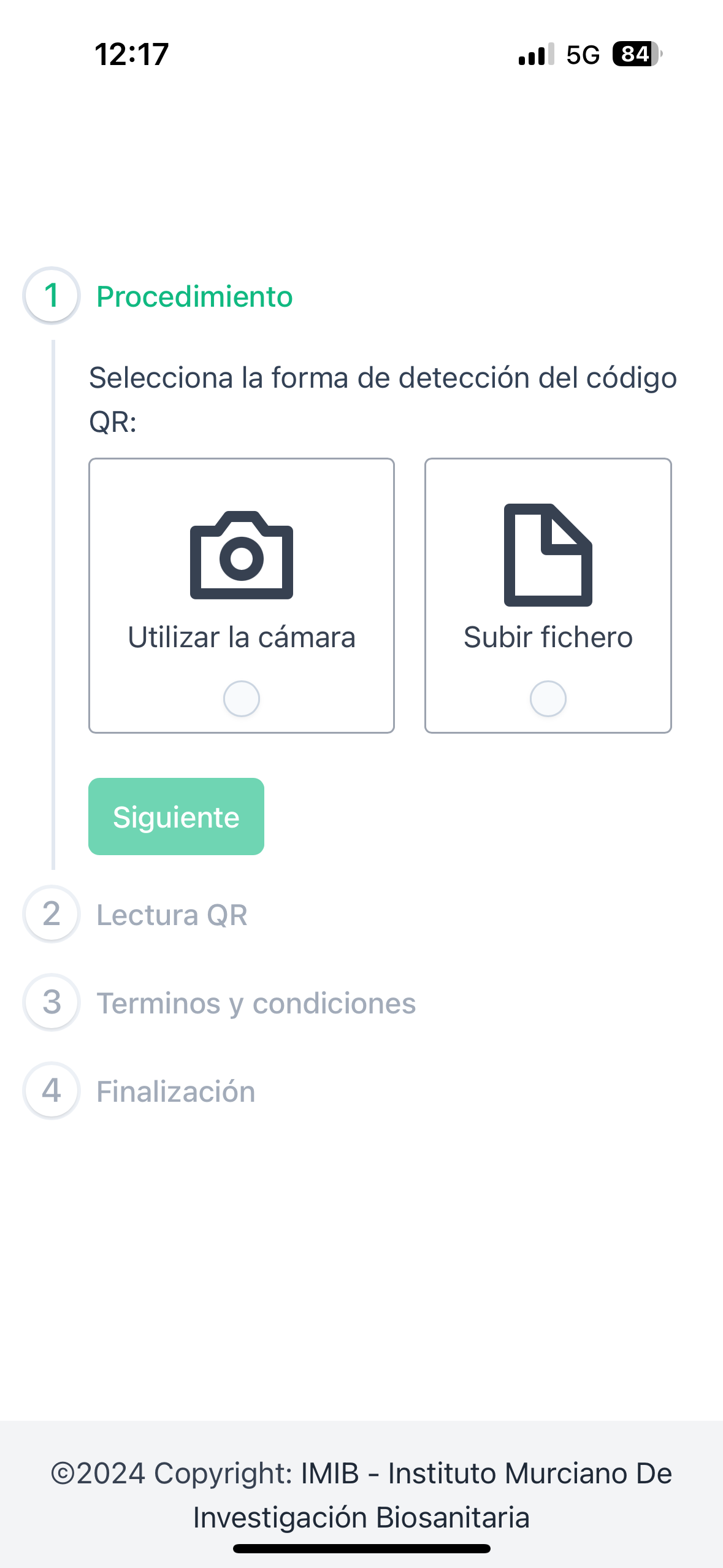
Instant registration & seamless access
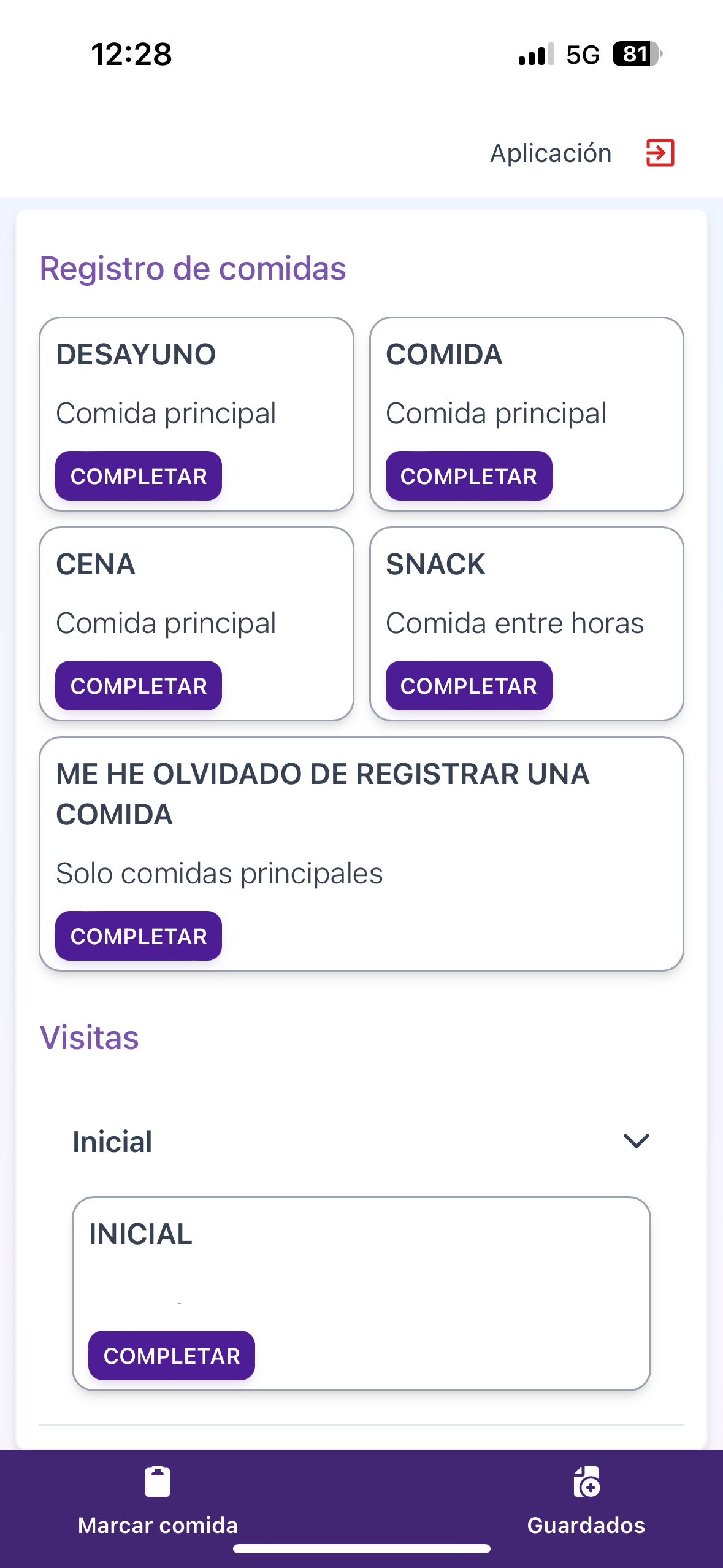
Browse tailored study instruments
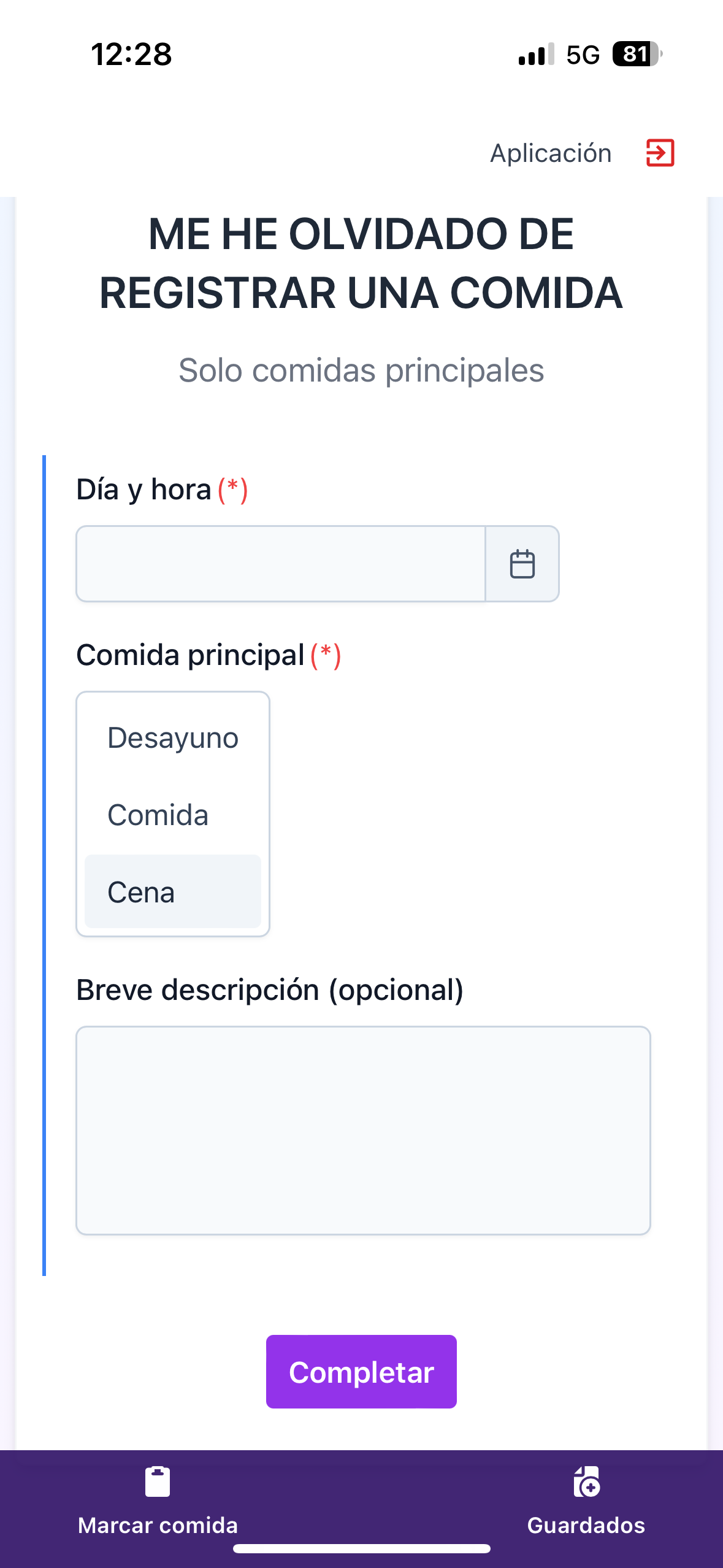
Easy & intuitive form completion
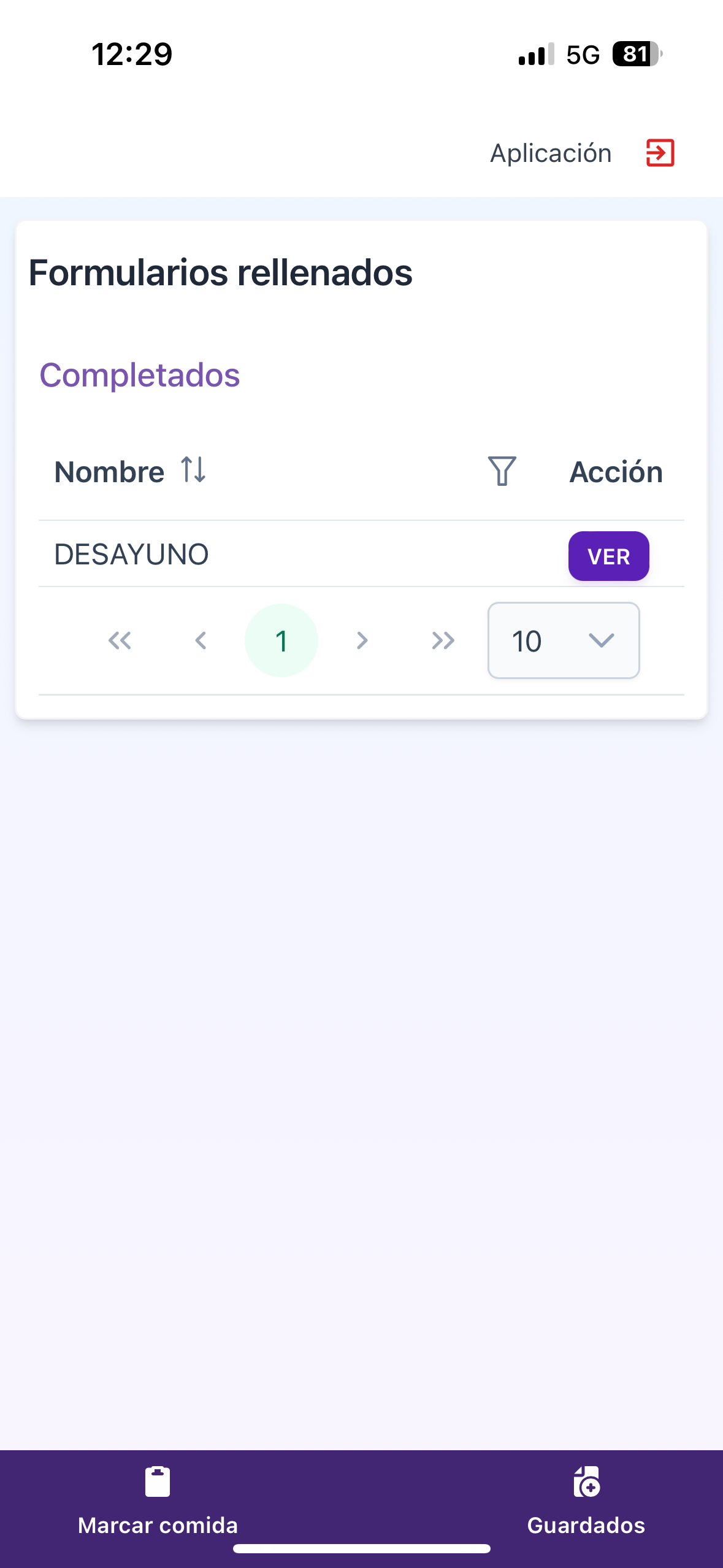
Clear view of completed questionnaires
Explore our semantic, scalable eCRFs and elevate your data management performance.
Request the service